
Separation of Plutonium from Uranium and Fission Products (1958)
U.S. Patent No. 2,855,269, granted on October 7, 1958, to George E. Boyd, Arthur W. Adamson, Jack Schubert, and Edwin R. Russell, describes a groundbreaking chemical process for isolating plutonium (Pu) from neutron-irradiated uranium. Filed during the height of the Manhattan Project in 1944, this patent was eventually assigned to the U.S. Atomic Energy Commission.
This invention solved one of the most difficult challenges of the nuclear age: how to extract minute quantities of plutonium (often only 0.02% of the total mass) from a mixture of uranium and highly radioactive fission products. Traditional chemical precipitation was messy and imprecise; this patent introduced ion-exchange chromatography to achieve high-purity separation.
The Innovation: Selective Adsorption
The process relies on “adsorption affinity”—the principle that different elements stick to specific resin surfaces with different levels of “strength.” By passing the radioactive mixture through columns of specialized resin, the scientists could force the elements to separate into distinct layers, or “strata.”
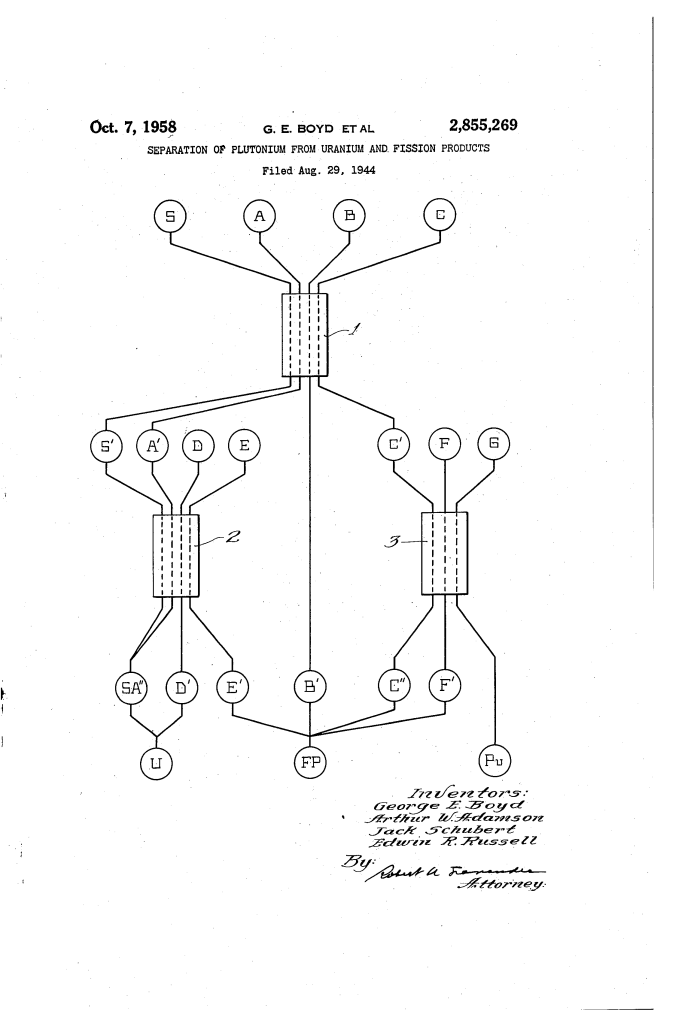
1. The Phenol-Formaldehyde Resin
The first stage uses a sulfonated organic resin. As the liquid uranium solution flows through the column, the plutonium and some fission products “exchange” ions with the resin and stick to it, while the bulk of the uranium passes through.
2. The Zirconium Phosphate Decontaminator
Plutonium is then washed off the first resin and passed through a second column containing zirconium phosphate. This material is highly selective; it “grabs” the plutonium values while allowing the remaining radioactive fission products to wash away, effectively “decontaminating” the sample.
How the Apparatus Functions
The separation is a multi-column process that acts like a chemical filter, refining the material at each step:
| Step | Action | Purpose |
| 1. Primary Adsorption | Irradiated uranium dissolved in nitric acid flows through Column 1. | Captures Plutonium (Pu) and fission products; discards the bulk of the Uranium. |
| 2. Uranium Wash | A dilute sulfuric acid wash (H_2SO_4) is passed through the resin. | Removes any lingering uranium without dislodging the plutonium. |
| 3. Plutonium Elution | A phosphoric/nitric acid mix (H_3PO_4 + HNO_3) is added. | Strips the plutonium and remaining fission products off the resin for the next stage. |
| 4. Decontamination | The eluate passes through Column 3 (Zirconium Phosphate). | Plutonium is preferentially adsorbed; “beta” and “gamma” radioactive waste passes through. |
| 5. Recovery | A concentrated 7M Nitric Acid wash is applied to the final column. | Recovers the purified Plutonium ($Pu$) in a concentrated liquid form. |
Technical Components
- Ion-Exchange Columns: Vertical tubes packed with finely divided resin (approx. 30–200 mesh). Smaller particles provide more surface area for faster adsorption.
- Sulfonated Resin: A hard, black organic resin containing sulfonic acid groups (R-SO_3-H) that attract metal cations.
- Zirconium Phosphate: A chemical compound prepared by digesting a precipitate in nitric acid at 90°C to 100°C to ensure it effectively “traps” plutonium ions.
- Eluate: The liquid gathered after it has passed through the adsorbent, containing the “stripped” values of the desired element.
Historical and Scientific Impact
This patent represents a major milestone in radiochemistry and the development of the nuclear fuel cycle.
- Purity Levels: By using two different types of adsorbents, the inventors achieved a separation efficiency where 91.2% of the plutonium was recovered with negligible radioactive contamination.
- Cold War Criticality: The ability to separate plutonium-239 was essential for the production of nuclear weapons and, later, the development of nuclear power reactors.
- Safety: This method allowed for “remote” chemical processing, reducing the direct exposure of scientists to the intense radiation of fission products.
About the Inventor: Edwin R. Russell
Edwin Roberts Russell was a distinguished African American chemist and a key member of the Manhattan Project at the University of Chicago’s Metallurgical Laboratory.
- Nuclear Pioneer: He was one of the few scientists tasked with the direct isolation of plutonium.
- Academic Excellence: Russell held a Ph.D. from the University of Chicago and later served as a professor at Howard University and a research scientist at the Savannah River Plant.
- Legacy: His work on this patent and others provided the chemical foundation for the safe handling and separation of radioactive isotopes, earning him a place among the most significant scientists of the atomic age.
Summary of Claims
The patent explicitly claims:
- The method of separating plutonium from uranium using phenol-formaldehyde cation exchange resins.
- The preferential adsorption of plutonium onto zirconium phosphate from an eluate containing nitric and phosphoric acids.
- The use of mineral acids (specifically 7M Nitric Acid) to remove and recover the purified plutonium from the final adsorbent.
- The sequential washing of resins to isolate “beta and gamma-active” fission products from the heavy metal values.
