Ion Exchange Absorption Process for Plutonium Separation (1961)
U.S. Patent No. 2,992,249, granted on July 11, 1961, to George E. Boyd, Edwin R. Russell, and Moddie D. Taylor, describes a specialized chemical process for isolating plutonium from neutron-irradiated uranium. This research was conducted at Oak Ridge National Laboratory under the auspices of the U.S. Atomic Energy Commission.
The challenge addressed by this patent was extreme: after uranium is irradiated in a nuclear reactor, it contains a mixture of uranium, plutonium, and highly radioactive “fission products” (like zirconium, columbium, and rare earths). Because these fission products are dangerously radioactive, a process was needed that was efficient, highly selective, and capable of being operated by remote control.
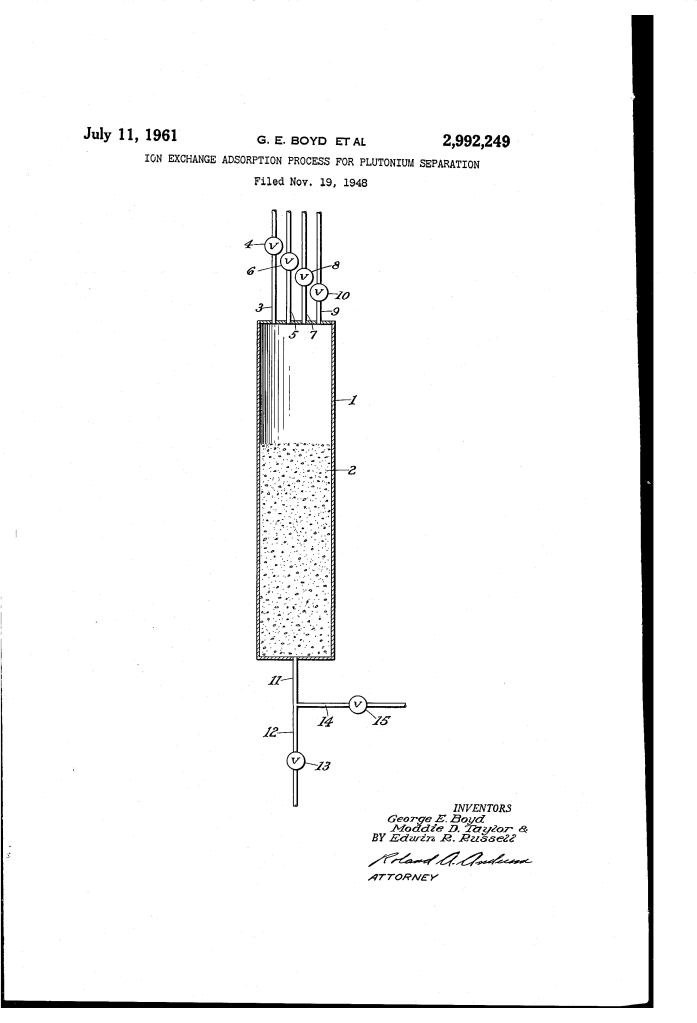
The Separation System
The invention utilizes a column-based ion-exchange system. In this setup, a solution is passed through a “bed” of adsorbent material that chemically traps specific ions.
1. The Adsorption Step
A solution of uranyl nitrate (containing plutonium and fission products) is fed into the column. The adsorbent—typically a phenol-formaldehyde resin containing sulfonic acid groups—acts like a chemical magnet, pulling the plutonium (Pu4+), uranyl ions (UO_2^2+), and various fission product cations out of the liquid and sticking them to the resin beads.
2. Selective Elution (The “Chemical Key”)
The brilliance of this patent lies in the use of specific carboxylic acids (such as oxalic, tartaric, or citric acid) to “unlock” and wash out specific elements one at a time. This is called selective elution.
- Step A: Uranium Removal: A 2.5% sulfuric acid solution is passed through to wash out the uranium.
- Step B: Fission Product Removal: A very dilute (0.75%) oxalic acid solution is used to wash away specific contaminants like zirconium and columbium. At this low concentration, the plutonium stays stuck to the resin.
- Step C: Plutonium Isolation: The concentration of the carboxylic acid is increased to 5%. This higher concentration forms a soluble complex with the plutonium, washing it out of the column in a highly pure state.
Key Innovations and Benefits
The Boyd-Taylor-Russell process offered several advantages over earlier precipitation methods:
| Feature | Pre-1961 Methods | The Boyd-Taylor Process |
| Method | Bulky chemical precipitation | Precision Ion Exchange |
| Control | Difficult to automate | Easily operated via Remote Control |
| Purity | Often contaminated with rare earths | High selectivity via concentration control |
| Waste | Large volumes of radioactive sludge | Concentrated, manageable liquid waste |
The Inventors: A Historical Note
Notably, Moddie Daniel Taylor was a prominent African American chemist and a member of the Manhattan Project. His work on this process was vital to the post-war development of nuclear technology. The ability to separate plutonium was essential not only for the defense industry but also for the development of “breeder” reactors and nuclear medicine.
Performance Data (Example I)
The patent provides specific results from a test run using a 100-day-old irradiated uranium mass:
- Adsorption: 100% of the plutonium was successfully trapped on the resin.
- Contaminant Wash (0.75% Oxalic Acid): Removed 42.3% of gamma radiation (mostly zirconium and columbium) while losing less than 2% of the plutonium.
- Plutonium Recovery (5% Oxalic Acid): Successfully recovered 95.4% of the original plutonium with extremely low radioactive contamination (only 1.5% of the original gamma activity).
Summary of Claims
The patent claims a method for recovering plutonium by:
- Adsorbing a solution onto a cation-exchange resin.
- Eluting contaminants with a dilute carboxylic acid (0.05% to 1%).
- Eluting the plutonium with a concentrated carboxylic acid (greater than 2%, ideally 5%).
- Utilizing specific acids like oxalic, tartaric, or citric to facilitate the selective complexing required for separation.
