High Silicon Cast Iron (1964)
U.S. Patent No. 3,129,095, granted on April 14, 1964, to Walter A. Luce, Glenn W. Jackson, Earl Ryder, and Mars G. Fontana, describes an advanced high-silicon alloy engineered to survive the most punishing chemical environments. Assigned to The Duriron Company of Dayton, Ohio, this invention revolutionized the production of chemical processing equipment and impressed current anodes used in maritime protection.
The innovation solved a “Holy Grail” problem in metallurgy: creating an affordable material that could resist both reducing acids (like hydrochloric) and oxidizing agents (like ferric chloride) at high temperatures without the prohibitive cost of “super-alloys” like nickel or titanium.
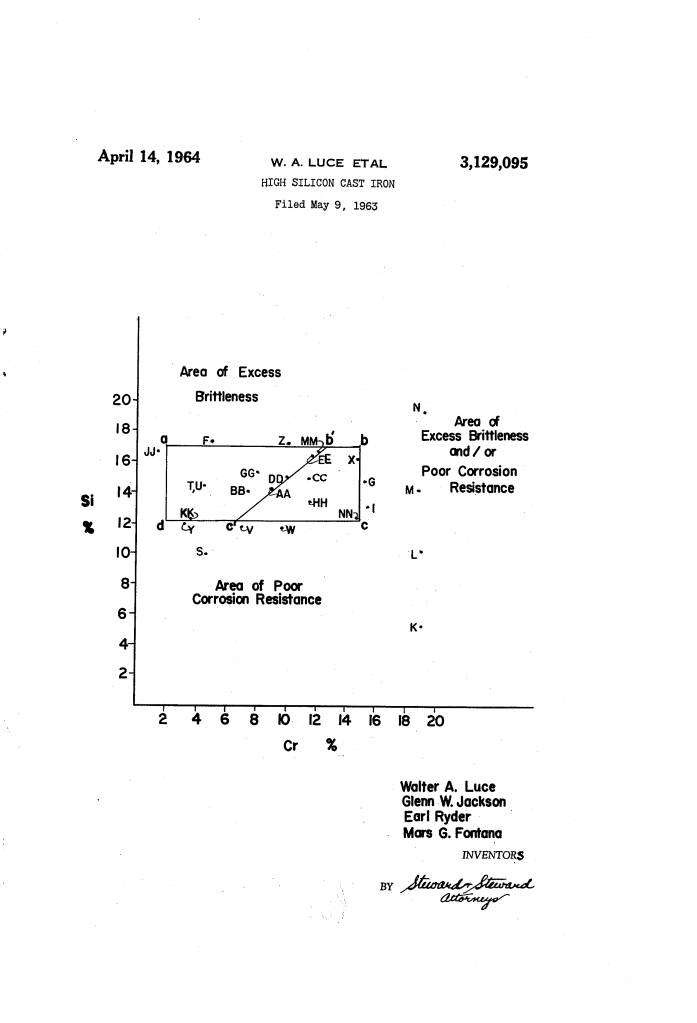
The Innovation: The “Chromium Paradox”
Historically, metallurgists avoided adding chromium to high-silicon iron because it was believed to increase corrodibility in reducing acids. The inventors discovered that if chromium was kept within a “critical limit,” it actually provided a startling leap in performance.
The “Cast Iron” Requirement
Unlike steels, these alloys must be true cast irons, meaning they contain free graphite randomly dispersed throughout the metal. This graphite is essential for:
- Mechanical Strength: Preventing the metal from being too brittle to handle.
- Thermal Shock Resistance: Allowing the metal to survive rapid temperature changes.
- Fluidity: Ensuring the molten metal can be cast into intricate industrial shapes like pump impellers or valves.
Key Chemical Components
The patent identifies a precise “sweet spot” where corrosion resistance and mechanical integrity meet.
| Component | Range (%) | Function |
| Silicon (Si) | 12% – 17% | Provides the primary “silico-ferrite” matrix that resists acid attack. |
| Chromium (Cr) | 2% – 15% | The “Key Addition.” Boosts resistance to chloride ions and high-temperature oxidation. |
| Molybdenum (Mo) | Up to 5% | Added for “extra-severe” service; specifically protects against boiling hydrochloric acid. |
| Carbon (C) | 0.1% – 3% | Must be high enough to form free graphite to prevent the alloy from becoming glass-brittle. |
| Boron (B) | Trace | Often added as a “grain refiner” to prevent the metal from spalling (chipping) during grinding. |
The “Anode” Breakthrough: Protecting Ships and Steel
One of the most significant applications for this alloy is as impressed current anodes. These are submerged metal rods that use electricity to prevent the hulls of ships or underground pipelines from rusting.
Performance in Synthetic Sea Water (93°C):
- Standard Silicon Iron: Suffered “selective attack,” pitting badly and falling apart.
- Luce’s Chromium Alloy: Experienced uniform consumption at a rate of less than 1 lb per ampere-year, making it economically superior and physically more durable.
Technical Performance: Mastering Hydrochloric Acid
The patent provides dramatic evidence of how these alloys outperform previous industry standards like Duriron and Durichlor when exposed to aggressive acids.
| Environment | Standard Silicon Iron (Mils/Year) | Invention Alloy (Mils/Year) |
| 20% Boiling HCl | Dissolved (Complete Failure) | 117 (Highly Resistant) |
| 20% HCl + Ferric Chloride | 1,600+ (Extreme Corrosion) | 34 (Excellent) |
| 40% Boiling Nitric Acid | 180 | 12 |
About the Inventors
The team represented a powerhouse of mid-century industrial chemistry and academia:
- Mars G. Fontana: A legendary figure in corrosion engineering from Ohio State University; the “Fontana Corrosion Center” is named in his honor.
- The Duriron Company: Based in Dayton, Ohio, they were the world leaders in high-silicon iron production.
- Legacy: This patent provided the chemical blueprint for modern “high-chrome” silicon irons used today in deep-well ground beds and chemical refineries.
Summary of Claims
The patent explicitly protects:
- An alloy containing 12% to 17% Silicon and 2% to 15% Chromium.
- The specific mathematical formula ($C = 1.66 – 0.063[Cr] – 0.089[Si]$) used to ensure enough carbon is present to form free graphite.
- The use of these alloys specifically for impressed current anodes with a transverse load capacity of at least 800 pounds.
