
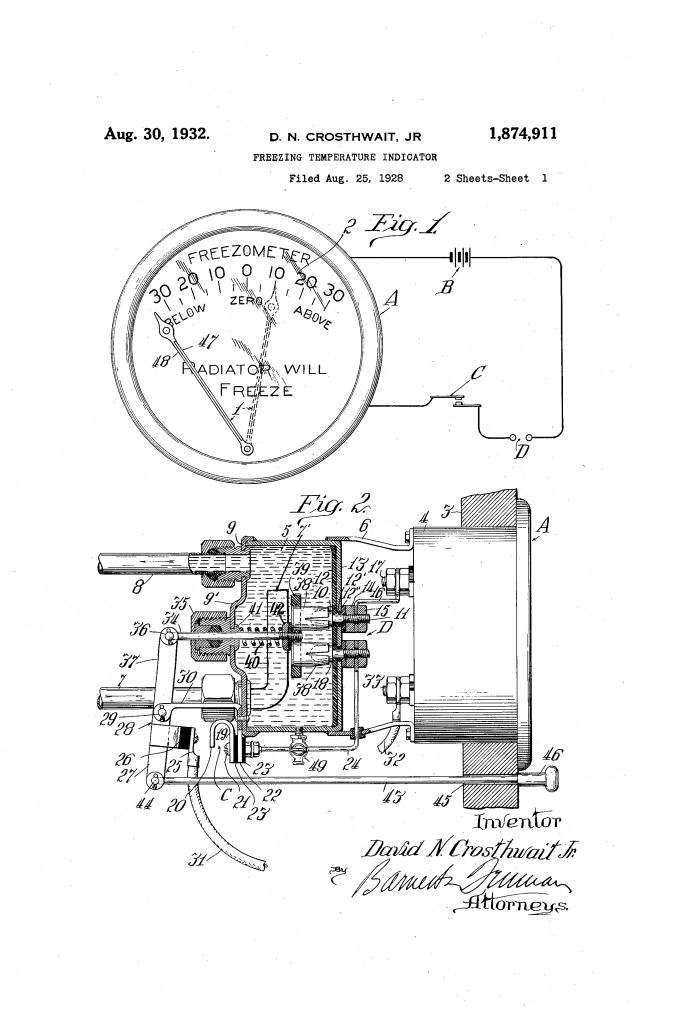
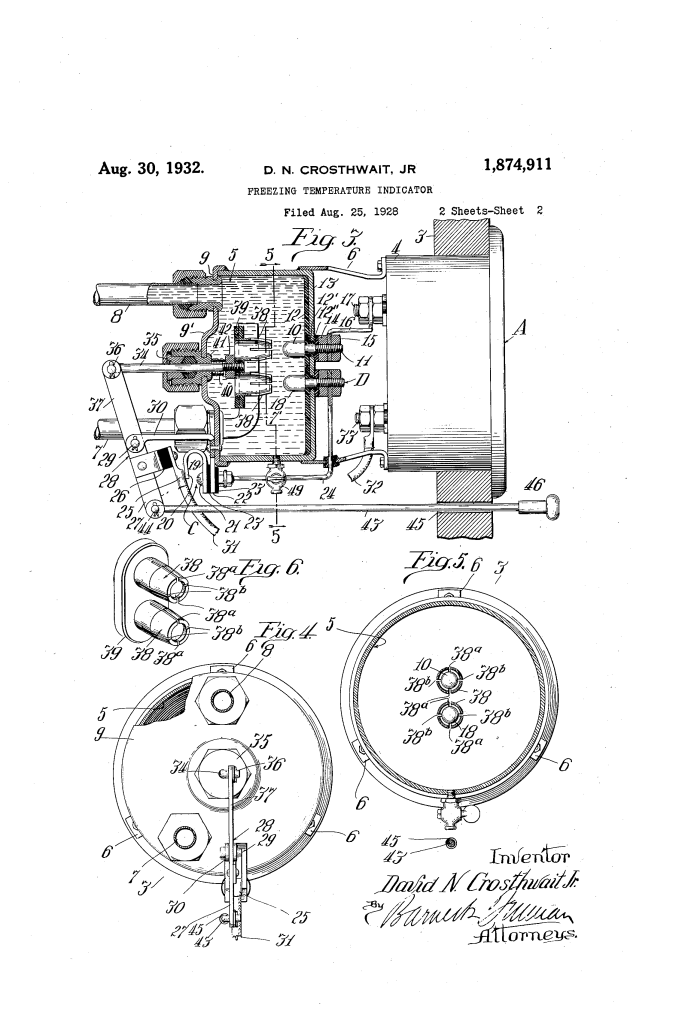
Freezing Temperature Indicator (David N. Crosthwait, Jr., No. 1,874,911)
This 1932 patent by David N. Crosthwait, Jr., a prolific African American engineer and pioneer in HVAC and electrical systems, describes a dashboard instrument for automobiles. At the time, drivers manually added anti-freeze (usually alcohol or glycerin) to radiators. Because these substances evaporate faster than water, drivers never knew the exact freezing point of their coolant without messy manual hydrometer tests.
Crosthwait’s invention provided an instant, dashboard-mounted reading of the coolant’s freezing point while the car was in motion.
1. The Electrical Conductivity Principle
The device operates on a simple scientific fact: the electrical resistance of a liquid changes depending on its chemical composition.
- The Circuit: An electrical current from the car’s battery (B) passes through a sample of the radiator fluid between two electrodes (D).
- Measuring Concentration: As the percentage of alcohol in the water increases, the solution’s resistance to electricity increases.
- The Galvanometer (A): The dash-mounted gauge is a sensitive electrical meter. Instead of showing “Volts” or “Amps,” its dial (2) is calibrated directly in degrees Fahrenheit. If the resistance is low (mostly water), the needle swings to 32 degrees F. If the resistance is high (more anti-freeze), the needle stays further to the left, indicating a much lower freezing point (e.g., 0 degrees F or -10 degrees F).
2. The Sampling Chamber and Circulation
To ensure the driver sees a “live” reading of the entire system, Crosthwait designed a sampling chamber (5) mounted behind the dashboard or under the hood.
- Continuous Flow: An inlet pipe (7) and outlet pipe (8) connect the chamber to the car’s cooling system. As the water pump moves coolant through the engine, a portion of it constantly flows through this small reservoir.
- Average Sample: Because the liquid is always circulating, the reading represents the average condition of the coolant, not just a stagnant pocket of water.
3. The Electrode Scraper Mechanism
A major challenge with early electrical sensors in water was incrustation—the buildup of minerals and chemicals on the electrodes that would give a false high-resistance reading.
- The Plunger (34): Crosthwait integrated a mechanical cleaning system. When the driver pulls a knob (46) on the dashboard to take a reading, it moves a lever.
- Spring Fingers (38): This lever pulls a set of tubular scrapers across the electrodes. These scrapers physically “shave” off any chemical deposits or rust.
- Sequence of Operation: The device is designed so that the scrapers finish their stroke before the switch (25) closes. This ensures the electrodes are perfectly clean at the exact moment the electrical measurement is taken.
4. Temperature Compensation
Because the conductivity of a liquid also changes based on how hot it is, Crosthwait included features to keep the gauge accurate even when the engine is running hot.
- Bi-Metallic Compensation: He proposed using a bi-metallic bar (47, 48) in the needle mechanism. These two metals expand at different rates when heated, causing the needle to “warp” or shift its base position to counteract the changes in the liquid’s conductivity caused by temperature.
Technical Component Summary
| Component | Function |
| Galvanometer (A) | The dashboard gauge that displays the freezing point. |
| Electrodes (10, 18) | Two metal rods that pass electricity through the coolant sample. |
| Scrapers (38) | Spring-tensioned tubes that clean the electrodes with every use. |
| Control Knob (46) | Manually pulled by the driver to clean electrodes and take a reading. |
| Return Spring (40) | Automatically retracts the scrapers and opens the switch when the knob is released. |
Engineering Significance
Crosthwait’s indicator was a precursor to modern automotive sensors. It replaced a difficult manual process with a “real-time” monitoring system. By combining mechanical cleaning with electrical measurement, he solved the problem of sensor degradation in harsh chemical environments—a principle still relevant in industrial fluid sensors today.
