
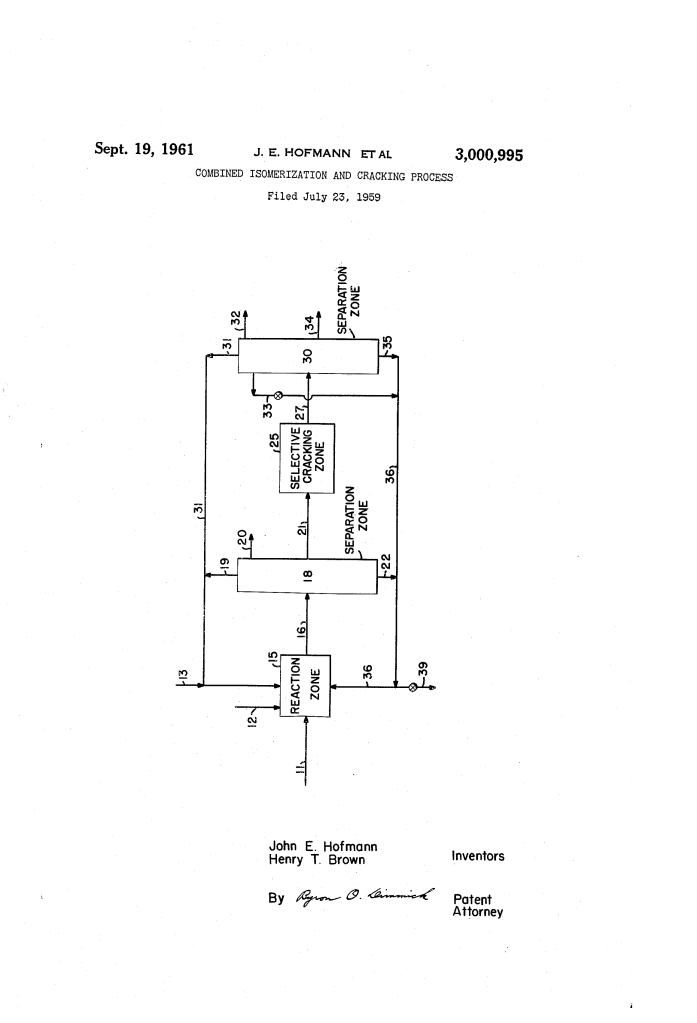
Combined Isomerization and Cracking Process (1961)
U.S. Patent No. 3,000,995, granted on September 19, 1961, to John E. Hofmann and Henry T. Brown, describes a chemical engineering process for upgrading low-quality petroleum fractions into high-performance motor fuels. Assignors to the Esso Research and Engineering Company, the inventors addressed the growing demand for high-antiknock (high-octane) fuels required by the high-compression engines of the 1960s.
The Chemical Problem: The “Heptane Headache”
In petroleum refining, isomerization is used to turn straight-chain hydrocarbons (like normal heptane) into branched-chain homologs (like dimethylpentane). Branched chains burn more smoothly in an engine, providing a higher octane rating.
However, standard isomerization of light naphtha (C_5 to C_7 hydrocarbons) results in an equilibrium mixture. While the C_5 and C_6 portions are high quality, the C_7 (Heptane) fraction typically contains 40–60% methylhexanes. These specific isomers have a relatively poor antiknock rating, dragging down the overall quality of the fuel.
The Solution: Selective Cracking
The core of this invention is a two-step process that doesn’t just isomerize the fuel, but “cleans up” the low-quality leftovers through selective cracking.
1. Primary Conversion (Isomerization/Alkylation)
The feed (light naphtha or a mix of butanes and heavier paraffins) is contacted with aluminum bromide (AlBr_3) at temperatures between 30°F and 140°F. This creates a mixture of branched-chain hydrocarbons.
2. Selective Cracking Step
The C_7 fraction is isolated and sent to a second reaction zone. Here, it is treated with a Friedel-Crafts catalyst (aluminum chloride or bromide) at a higher temperature (150°F to 250°F).
- The Selective Action: At this specific temperature range, the low-quality methylhexanes are unstable and “crack” into smaller, high-quality molecules like isobutane, isopentane, and isohexane.
- The Preservation: High-quality C_7 isomers, such as trimethylbutane and dimethylpentane, remain undisturbed.
Process Flow Summary
The patent outlines a circular efficiency model:
- Separation: Products from the first stage are fractionated.
- Upgrading: The C_7 cut goes to the cracking zone.
- Recycling: The resulting smaller isomers (butanes and pentanes) are recycled back to the first stage to serve as feed for further alkylation, maximizing the yield of high-octane components.
Comparative Performance (Example I)
The patent includes experimental data showing the transformation of the C_7 cut:
| Component | Original Feed | After Selective Cracking |
| Methylhexanes (Low Octane) | ~44% | Reduced to ~15% |
| Dimethylpentanes (High Octane) | ~29% | Maintained at ~28% |
| Isobutane/Isopentane (Byproducts) | 0% | Increased (Recyclable) |
Results based on aluminum chloride catalyst at 170°F.
Technical Specifications
| Parameter | Value/Range |
| Isomerization Temp | 30°F – 140°F |
| Cracking Temp | 150°F – 250°F (Preferred: 160°F – 190°F) |
| Primary Catalyst | Aluminum Bromide (AlBr_3) |
| Cracking Catalyst | AlCl_3 or AlBr_3 (often on Porocel support) |
| Antiknock Goal | Reduction of methylhexanes in C_7 fraction |
