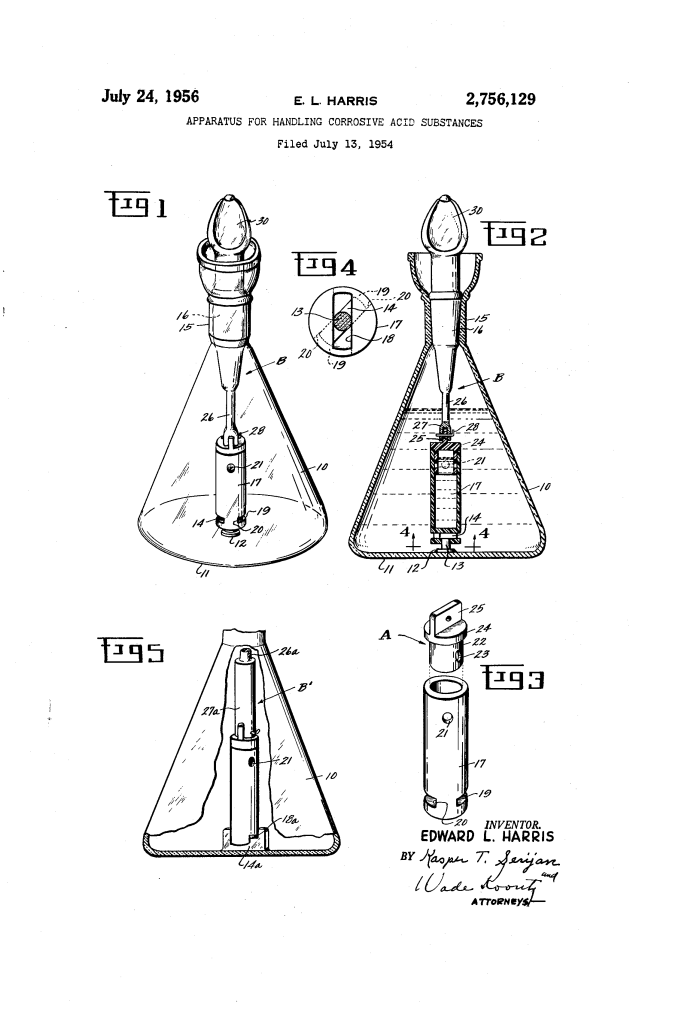
Apparatus for Handling Corrosive Acid Substances (1956)
U.S. Patent No. 2,756,129, granted on July 24, 1956, to Edward L. Harris, addresses a critical safety and equipment problem in chemical laboratories: the analysis of “super-corrosives” like hydrofluoric acid and fuming nitric acid.
These acids are so aggressive that they dissolve glass and stainless steel almost instantly. Traditionally, chemists had to use expensive platinum flasks for these reactions. Harris, an inventor at Wilberforce, Ohio, developed a way to use standard, inexpensive glass laboratory equipment to neutralize these acids by keeping the acid completely isolated from the glass until the moment it is neutralized.
The Core Design: The “Vial-in-a-Flask” System
The brilliance of the Harris apparatus is its sealed, externally operated transition. It allows a chemist to introduce a deadly acid into a neutralizing base (like sodium hydroxide) without any “free acid” ever touching the glass walls of the main flask.
1. The Corrosion-Resistant Inner Vial (17)
The heart of the system is a small weighing vial made from a haloethylenic polymer (modernly known as Teflon or PTFE).
- This material is immune to the “eating” action of hydrofluoric acid.
- The vial contains the acid sample and is sealed with a rotatable hollow plug (22).
2. The Key and Bayonet Lock (14, 18)
To operate the internal valve from the outside, the vial must be held perfectly still at the bottom of the flask.
- Harris installed a mounting base (12) with a rectangular key (14) at the bottom of the glass flask.
- The bottom of the Teflon vial has a bayonet slot (18). The chemist drops the vial onto the key and twists it 45 degrees to lock it to the floor of the flask.
How the Apparatus Functions
The process follows a strict sequence to ensure the safety of the operator and the equipment:
| Step | Action | Safety Purpose |
| 1. Charging | Acid is placed in the Teflon vial; the plug is rotated to close the ports (21, 23). | Keeps the acid safe during weighing and transport. |
| 2. Assembly | The vial is pinned to an actuating shaft (26) and lowered into a glass flask filled with alkaline solution. | The alkali acts as a “protective bath” for the glass. |
| 3. Sealing | A tapered stopper (16) on the shaft seals the neck of the glass flask. | Creates a closed system to contain toxic fumes or heat from the reaction. |
| 4. Activation | The chemist turns the external knob (30). This aligns the ports in the vial. | The acid flows out directly into the alkali, neutralizing instantly. |
Technical Components
- Reaction Flask (10): Standard glass vessel. Because it only ever touches the neutralized liquid, it does not corrode.
- Actuating Means (B): A long shaft that connects the external handle to the internal valve. This allows the chemist to stay a safe distance from the chemical interface.
- Neutralizing Zone: The area surrounding the vial. By keeping an excess of alkali in this zone, Harris ensures that the pH never drops low enough to damage the glass.
Historical and Scientific Impact
Edward L. Harris’s invention was particularly important for government and military research (the patent was “Granted under Title 35,” meaning the government could use it royalty-free).
- Cost Reduction: It eliminated the need for platinum labware, which was (and is) prohibitively expensive.
- Safety: Before this, transferring volatile acids into neutralization baths often resulted in “spattering” or the release of dangerous hydrogen fluoride gas. This “sealed system” kept all vapors inside.
- Analytical Precision: Because the vial can be weighed before and after assembly, chemists could achieve much higher accuracy in determining the concentration of corrosive substances.
About the Inventor: Edward L. Harris
Edward L. Harris was a scientist at Central State University (formerly College) in Wilberforce, Ohio—a historically Black university (HBCU). His work in the mid-1950s contributed significantly to the field of analytical chemistry, specifically in handling materials that were critical to the budding aerospace and nuclear industries of the Cold War era.
